
So it is usually fairly obvious if you have done something wrong, and you can check back again. If you get this wrong, you are going to end up with a silly answer, out by a factor of a thousand or a million. Similarly, if you are working out a volume using the equation, remember to covert the answer in cubic metres into dm 3 or cm 3 if you need to - this time by multiplying by a 1000 or a million. You would have to divide a volume in dm 3 by 1000, or in cm 3 by a million. So if you are inserting values of volume into the equation, you first have to convert them into cubic metres. That's because the SI unit of volume is the cubic metre, m 3 - not cm 3 or dm 3. This is the most likely place for you to go wrong when you use this equation. Should you want to convert from other pressure measurements: 
You must make that conversion before you use the ideal gas equation. These mean exactly the same thing.īe careful if you are given pressures in kPa (kilopascals). Pressure is measured in pascals, Pa - sometimes expressed as newtons per square metre, N m -2. 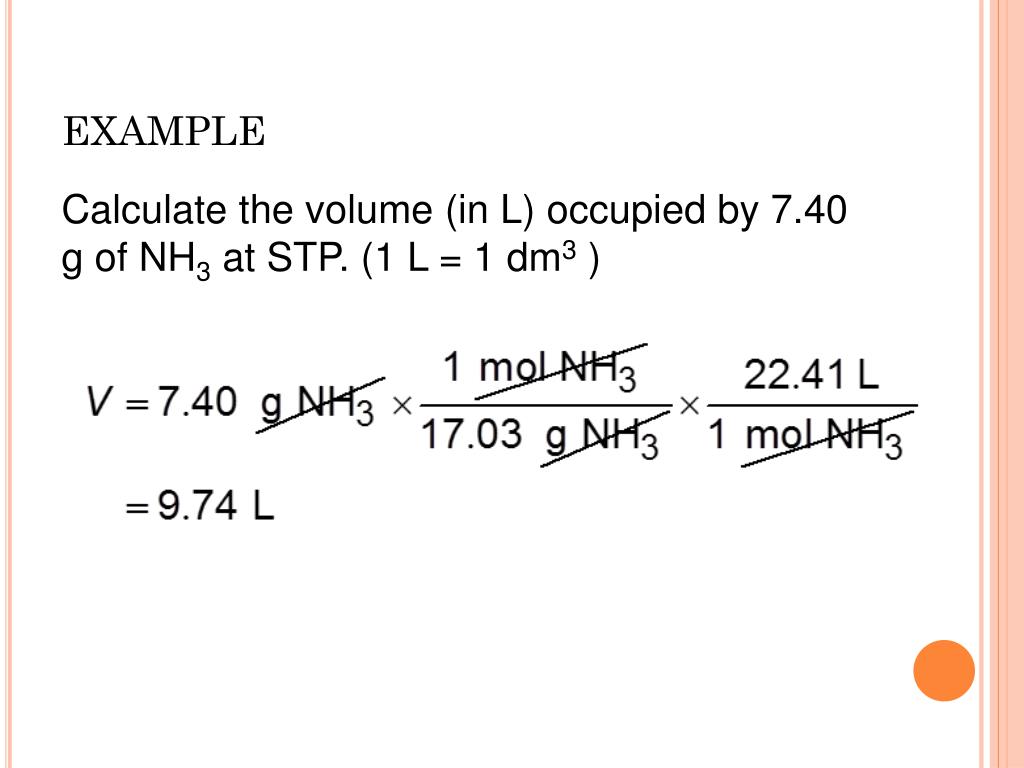
I am assuming below that you are working in strict SI units (as you will be if you are doing a UK-based exam, for example). The problems lie almost entirely in the units. On the whole, this is an easy equation to remember and use. The volume occupied by the molecules themselves is entirely negligible relative to the volume of the container. There are no (or entirely negligible) intermolecular forces between the gas molecules. The temperature of the gas is proportional to the average kinetic energy of the molecules.Īnd then two absolutely key assumptions, because these are the two most important ways in which real gases differ from ideal gases: (That means that there is no loss of kinetic energy during the collision.) 
Pressure is due to collisions between the molecules and the walls of the container.Īll collisions, both between the molecules themselves, and between the molecules and the walls of the container, are perfectly elastic. Gases are made up of molecules which are in constant random motion in straight lines. Real gases are dealt with in more detail on another page. There is no such thing as an ideal gas, of course, but many gases behave approximately as if they were ideal at ordinary working temperatures and pressures. Kinetic Theory assumptions about ideal gases This is intended only as an introduction suitable for chemistry students at about UK A level standard (for 16 - 18 year olds), and so there is no attempt to derive the ideal gas law using physics-style calculations. This page looks at the assumptions which are made in the Kinetic Theory about ideal gases, and takes an introductory look at the Ideal Gas Law: pV = nRT. This makes for a very useful approximation: any gas at STP has a volume of 22.4 L per mole of gas that is, the molar volume at STP is 22.4 L/mol (Figure 6.3 “Molar Volume”).Ideal gases and the ideal gas law: pV = nRT Standard temperature and pressure (STP) is defined as exactly 100 kPa of pressure (0.986 atm) and 273 K (0☌). The standard temperature is 273 K (0 degrees Celsius or 32 degrees Fahrenheit), and the standard pressure is 1 atm. STP is most widely used in measurements involving fuels, such as gas density. The abbreviation STP stands for Standard Temperature and Pressure in chemistry. Note that this form specifically stated 0 o C degree, not 273 Kelvin, even thought you will have to convert into Kelvin when plugging this value into the Ideal Gas equation or any of the simple gas equations. The universal value of STP is 1 atm (pressure) and 0 o C. Now, we have got the complete detailed explanation and answer for everyone, who is interested! This is a question our experts keep getting from time to time.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
